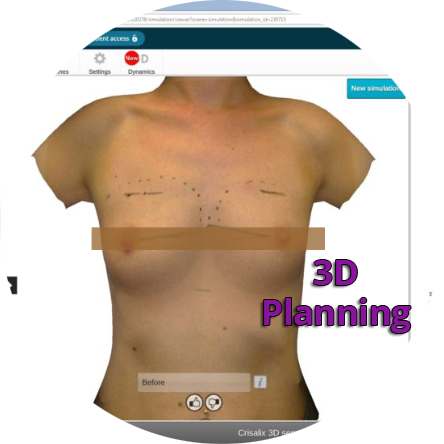
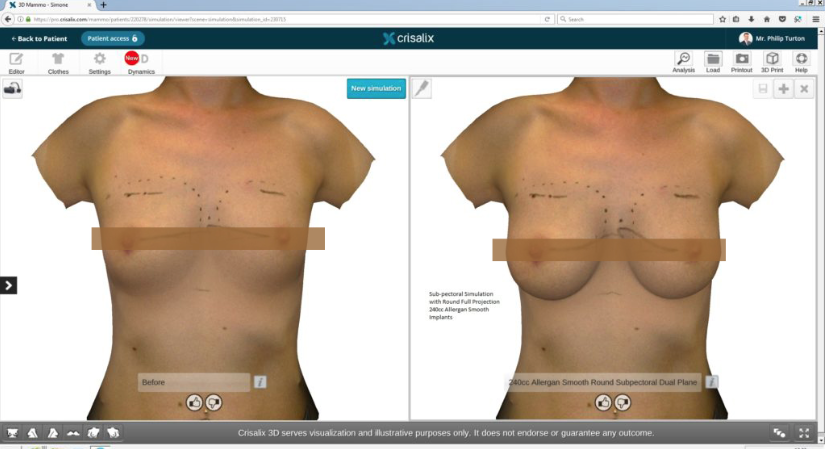
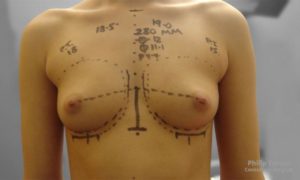
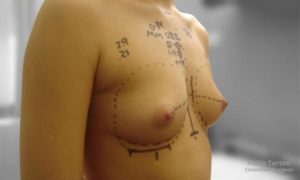
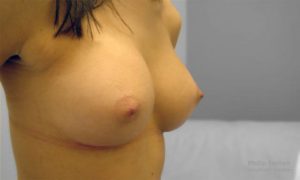
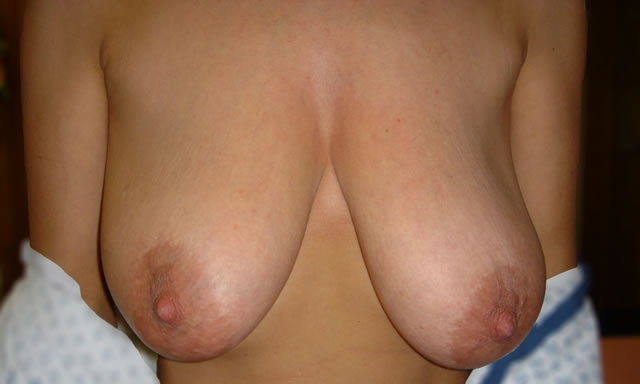
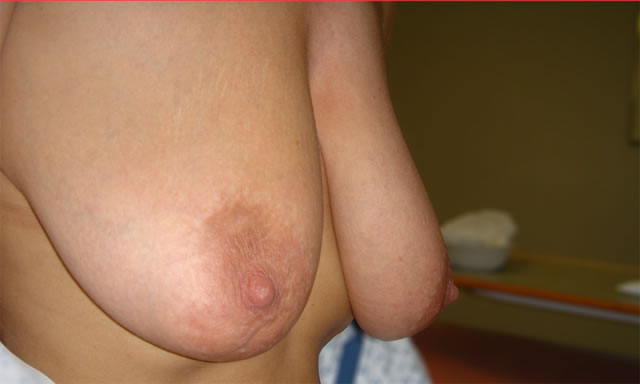
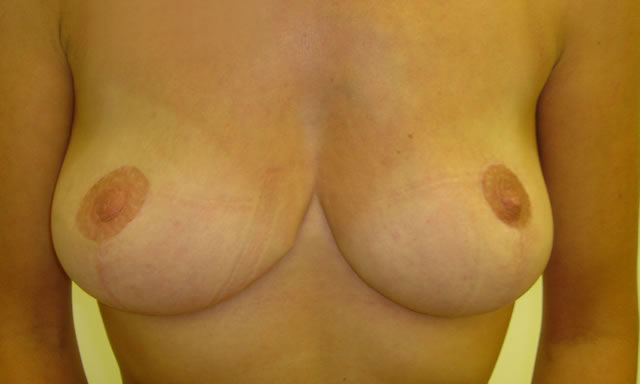
Initial Consultation Enquiries
All Private Enquiries are handled in confidence by Mr Turton’s PA: Victoria Short
You have a choice to see Mr Turton at either the Leeds Nuffield Hospital or the Spire Hospital Leeds
Please note that after an out-patient consultation and any surgery, in line with medical recommendations and the vast majority of patient’s own preferences, a letter is sent to your GP as a record. Please email Victoria Short with explicit instructions if this is something you wish not to occur, and also make it clear at the consultation with written instructions for Mr Turton’s medical file.
Please fill out the basic initial enquiry form, or preferably use the more detailed enquiry form on the bookings page of this website. If you need to direct your questions differently, please feel free to get in touch with Victoria Short by email to ensure that we can deal with your consultation in the most effective and efficient manner possible.